
Tetrahedron Letters | Vol 43, Issue 2, Pages 177-345 (7 January 2002) | ScienceDirect.com by Elsevier

Tetrahedron Letters | Vol 57, Issue 19, Pages 2017-2132 (11 May 2016) | ScienceDirect.com by Elsevier

Application of Fischer Indolization under Green Conditions using Deep Eutectic Solvents - Kotha - 2017 - The Chemical Record - Wiley Online Library

Structural Transformation and Second-Harmonic-Generation Activity in Rare-Earth and d0 Transition-Metal Oxysulfides RE3NbS3O4 (RE = Ce, Sm, Gd, Dy) | Inorganic Chemistry

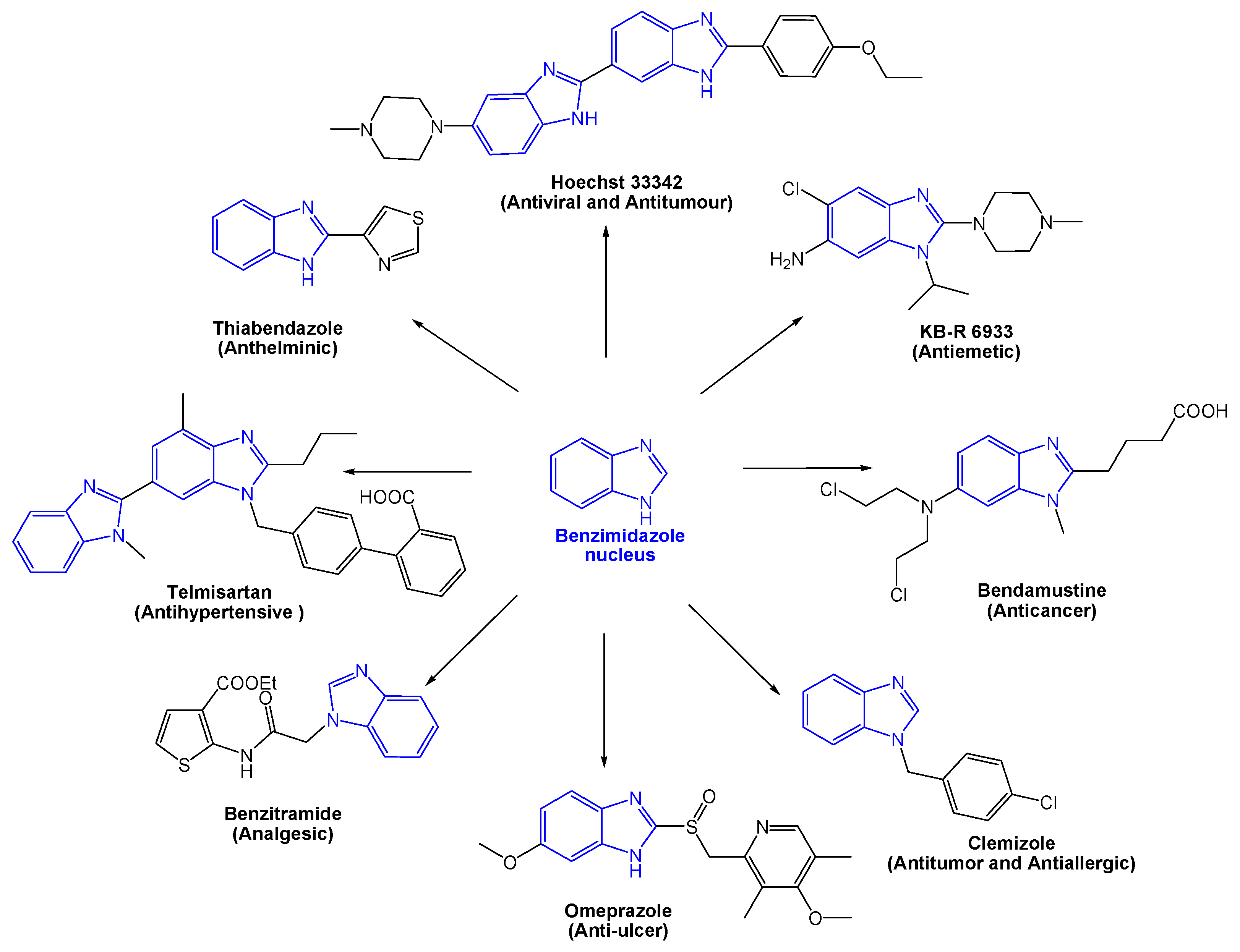
Molecules | Free Full-Text | Green Synthesis of Privileged Benzimidazole Scaffolds Using Active Deep Eutectic Solvent

Tetrahedron Letters | Vol 50, Issue 10, Pages 1087-1152 (11 March 2009) | ScienceDirect.com by Elsevier

A mild method for the conversion of activated aryl methyl groups to carboxaldehydes via the uncatalyzed periodate cleavage of enamines - ScienceDirect

Vinylphosphonium Salt-Mediated Reactions: A One-Pot Condensation Approach for the Highly cis-Selective Synthesis of N-Benzoylaziridines and the Green Synthesis of 1,4,2-Dioxazoles as Two Important Classes of Heterocyclic Compounds | Organic Letters
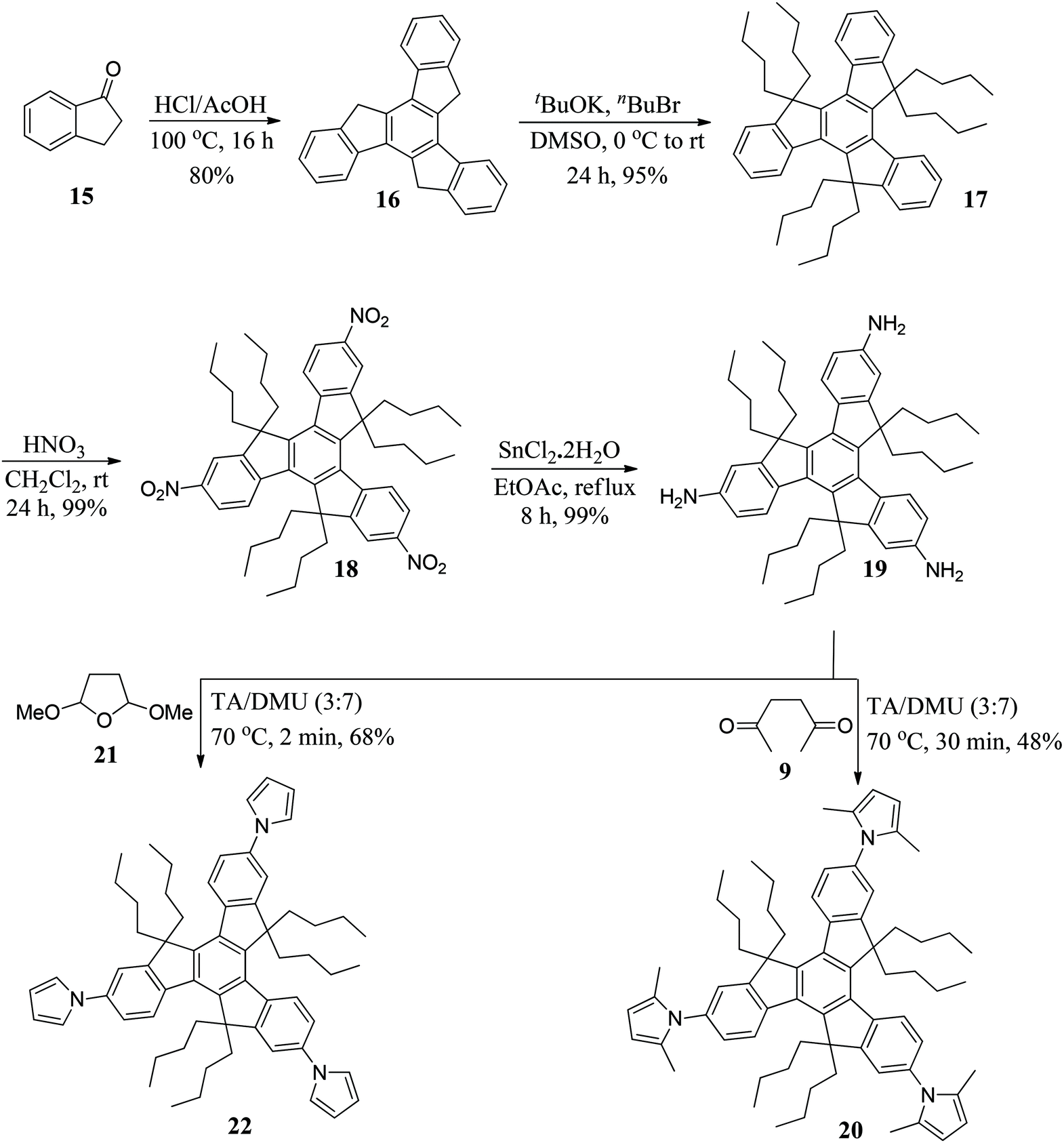
An expeditious and highly efficient synthesis of substituted pyrroles using a low melting deep eutectic mixture - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB01618K
An expeditious and highly efficient synthesis of substituted pyrroles using a low melting deep eutectic mixture - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D1OB01618K

Tetrahedron Letters | Vol 43, Issue 2, Pages 177-345 (7 January 2002) | ScienceDirect.com by Elsevier
![Palladium‐Catalyzed Decarboxylative Synthesis of 5H‐Benzo[4,5][1,3]oxazino[2,3‐a]isoindole‐5,11(6aH)‐Diones using 2‐Phenyl‐4H‐Benzo[d][1,3]oxazin‐4‐Ones and α‐Oxo Carboxylic Acids - Lalji - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library Palladium‐Catalyzed Decarboxylative Synthesis of 5H‐Benzo[4,5][1,3]oxazino[2,3‐a]isoindole‐5,11(6aH)‐Diones using 2‐Phenyl‐4H‐Benzo[d][1,3]oxazin‐4‐Ones and α‐Oxo Carboxylic Acids - Lalji - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/c212c9b9-c949-49b9-8c3b-0a931760d7f5/adsc201901142-fig-5001-m.jpg)
Palladium‐Catalyzed Decarboxylative Synthesis of 5H‐Benzo[4,5][1,3]oxazino[2,3‐a]isoindole‐5,11(6aH)‐Diones using 2‐Phenyl‐4H‐Benzo[d][1,3]oxazin‐4‐Ones and α‐Oxo Carboxylic Acids - Lalji - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Catalysis in Cyclodextrin-Based Unconventional Reaction Media: Recent Developments and Future Opportunities | ACS Sustainable Chemistry & Engineering

PDF) Planar force-constant method for lattice dynamics of cubic III-nitrides | HORACIO WAGNER LEITE ALVES - Academia.edu

Tetrahedron Letters | Vol 39, Issue 8, Pages 727-918 (19 February 1998) | ScienceDirect.com by Elsevier

An investigation into the unusual linkage isomerization and nitrite reduction activity of a novel tris(2-pyridyl) copper complex | Royal Society Open Science

Vinylphosphonium Salt-Mediated Reactions: A One-Pot Condensation Approach for the Highly cis-Selective Synthesis of N-Benzoylaziridines and the Green Synthesis of 1,4,2-Dioxazoles as Two Important Classes of Heterocyclic Compounds | Organic Letters

Ultimate Manipulation of Magnetic Moments in the Golden Tetrahedron Au20 with a Substitutional 3d Impurity | The Journal of Physical Chemistry C
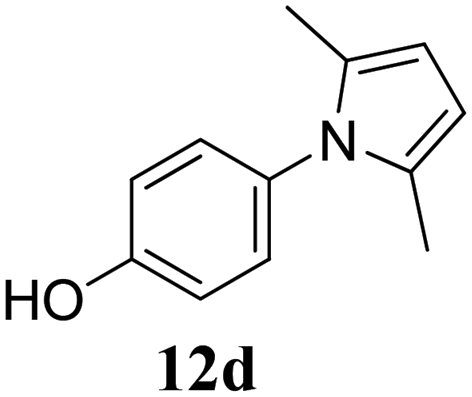
Molecules | Free Full-Text | Green Synthesis of Privileged Benzimidazole Scaffolds Using Active Deep Eutectic Solvent
![Palladium‐Catalyzed Decarboxylative Synthesis of 5H‐Benzo[4,5][1,3]oxazino[2,3‐a]isoindole‐5,11(6aH)‐Diones using 2‐Phenyl‐4H‐Benzo[d][1,3]oxazin‐4‐Ones and α‐Oxo Carboxylic Acids - Lalji - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library Palladium‐Catalyzed Decarboxylative Synthesis of 5H‐Benzo[4,5][1,3]oxazino[2,3‐a]isoindole‐5,11(6aH)‐Diones using 2‐Phenyl‐4H‐Benzo[d][1,3]oxazin‐4‐Ones and α‐Oxo Carboxylic Acids - Lalji - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/754f9321-3a46-441c-aa76-3de9ccdb6254/adsc201901142-fig-5003-m.jpg)
Palladium‐Catalyzed Decarboxylative Synthesis of 5H‐Benzo[4,5][1,3]oxazino[2,3‐a]isoindole‐5,11(6aH)‐Diones using 2‐Phenyl‐4H‐Benzo[d][1,3]oxazin‐4‐Ones and α‐Oxo Carboxylic Acids - Lalji - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library





